
Water
Quality Assessment: Chemical: pH
The most commonly
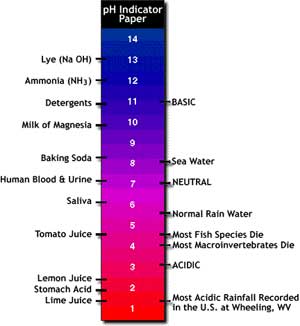
measured chemical attribute of water is its acidity or pH. The pH scale
ranges from 1 to 14, with 1 being the most acidic and 14 being the most
basic. Most streams have a neutral to slightly basic pH of 6.5 to 8.5.
If streamwater has a pH less than 5.5, it may be too acidic for fish to
survive in, while streamwater with a pH greater than 8.6 may be too basic.
Image: a pH strip with the values of common
substances labeled.
A change in streamwater pH can also affect aquatic life indirectly by altering other aspects of water chemistry. For example, low pH levels can increase the solubility of certain heavy metals. This allows the metals to be more easily absorbed by aquatic organisms.
A shift of pH in either direction from neutral may indicate the presence of a pollutant in the stream. However, since some streams are naturally acidic — or basic — pH may not necessarily indicate pollution. The pH of clean water depends on several factors, including the types of rock and vegetation within the watershed. The water that enters a stream passes through the canopy of the trees, percolates through the soil, and flows through the groundwater system to eventually empty into the stream. During its travels, the water weathers the rocks and soils and picks up acidic and alkaline (high pH) compounds throughout the watershed. For example, streams draining forests and marshes are often slightly acidic due to the presence of humic acids produced by decaying vegetation in the soil. On the other hand, streams in watersheds situated on limestone (CaCO3) contain high concentrations of bicarbonate ions. This results in alkaline waters.
Alkaline water can act as a weak buffer solution, depending on the concentrations of carbonates and bicarbonates. Therefore an acidic pollutant may be present in such water, yet not cause a change in the stream's pH. For this reason, alkalinity should be examined in conjunction with pH.
Overview ..|.. Biological Assessment ..|.. Chemical Assessment ..|.. Physical Assessment.
pH
/ Alkalinity / Hardness
/ Nitrates. Nitrites, and Ammonia / Ortho-
and Total Phosphate / Dissolved Oxygen and
Biochemical Oxygen Demand / Fecal Coliform
/ Conductivity and Density
Glossary .|.
Related Links
.|..
References
..|..
PBL Model
.|
Home ..|.. Teacher Pages ..|.. Modules & Activities
HTML code by Chris Kreger
Maintained by ETE Team
Last updated November 10, 2004
Some images © 2004 www.clipart.com
Privacy Statement and Copyright © 1997-2004 by Wheeling Jesuit University/NASA-supported Classroom of the Future. All rights reserved.
Center for Educational Technologies, Circuit Board/Apple graphic logo, and COTF Classroom of the Future logo are registered trademarks of Wheeling Jesuit University.
